Medical device testing outsourcing to hit $15.3Bn by 2034: Market.us
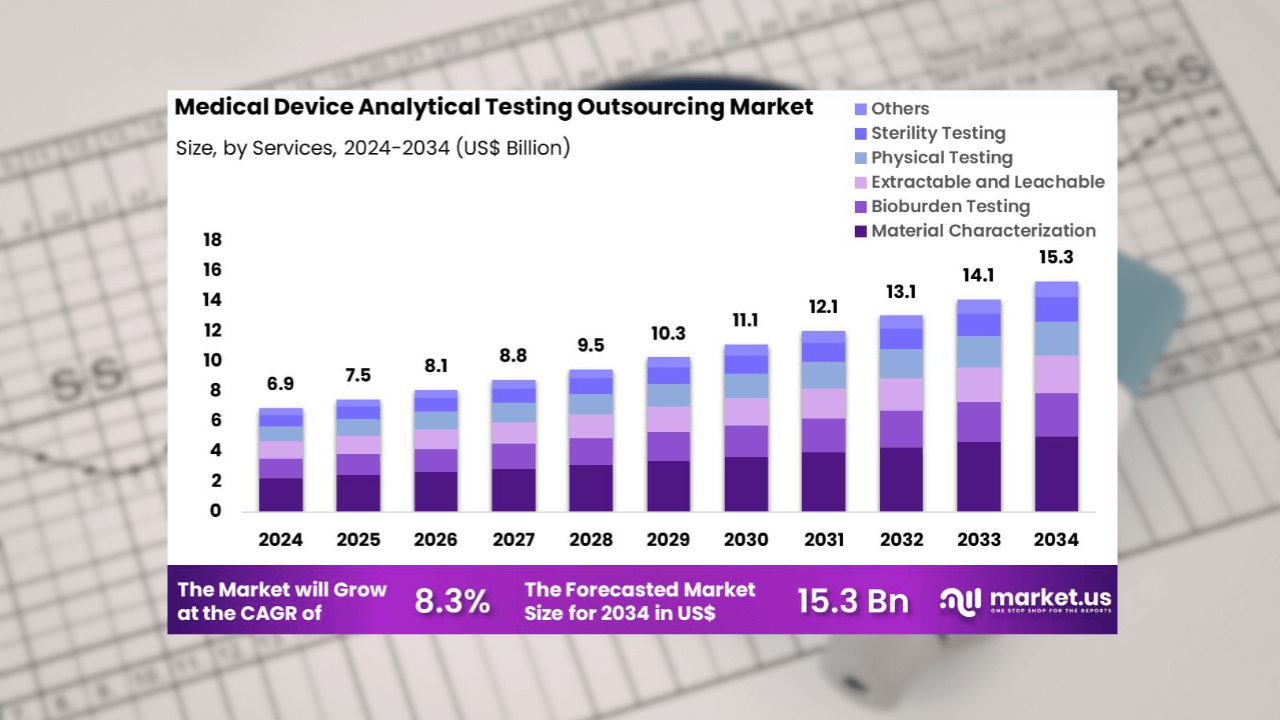
NEW YORK, UNITED STATES — The global medical device analytical testing outsourcing market is projected to more than double over the next decade, reaching $15.3 billion by 2034 from $6.9 billion in 2024, according to a recent report by Market.us.
Organizations experience growth acceleration as they need to meet increasing regulatory requirements while developing their specialized testing capabilities for cardiology and hospital-based testing.
Material characterization leads services growth
Outsourced testing services are increasingly crucial for manufacturers facing complex device designs.
“Material characterization contributed 32.8% of growth within services and led the medical device analytical testing outsourcing market due to rising complexity in device materials and design,” the study noted.
Laboratories conduct comprehensive chemical and physical tests to evaluate advanced polymers, alloys, and composite materials, which helps determine their performance and safety standards.
Services also include bioburden testing, extractable and leachable analysis, sterility testing, and physical evaluations that support accelerated product development cycles.
Cardiology became the leading therapeutic area, which produced 29.6% of the total growth. The testing process needs to assess stents, catheters, and implantable monitors as the devices need to prove their material strength according to safety regulations.
The report emphasized that manufacturers depend more on outsourcing services to achieve their regulatory requirements while they continue to accelerate their product development process.
“Frequent design updates and next-generation cardiac devices drive repeated testing cycles,” Market.us added, highlighting the ongoing demand for specialized analytical support.
Hospitals remain top end‑users as outsourcing rises
Hospitals remain the largest end-users of outsourced medical device testing, representing 47.9% of the market.
Their involvement in clinical validation, pilot studies, and post-market surveillance demonstrates how crucial analytical testing is to protect patient safety.
Large teaching hospitals and health systems frequently collaborate with manufacturers, increasing demand for specialized laboratories.
“Expansion of hospital-based research programs further supports growth,” the study noted, reflecting a strong institutional reliance on external testing capabilities.
North America leads the market with a 39.4% share, driven by regulatory requirements and a concentration of medtech hubs. At the same time, Asia Pacific will achieve its highest growth rate during the forecast period as China, India, and South Korea expand their testing infrastructure and implement automation for compliance.
Outsourcing partners bring AI technology, robotic systems, and eco-friendly testing approaches into their operations as these advanced technologies deliver better operational results and more accurate forecasting capabilities.
Expansion for medical device analytical testing will continue to grow due to devices getting more complicated, as well as demanding international regulations.
The sector will benefit from close collaborations between manufacturers, hospitals, and specialized laboratories in consolidating its role as a major keystone in device safety, innovation, and rapid market penetration.

 Independent
Independent




